
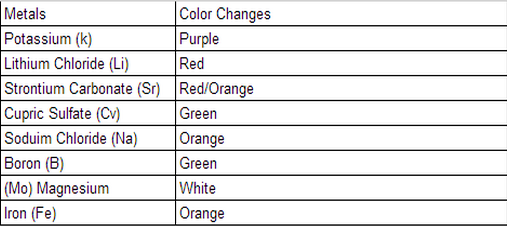
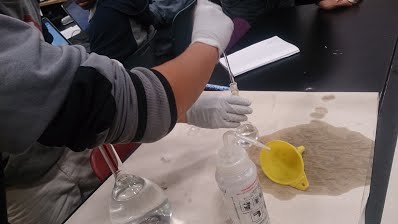
For this lab we took different metals from the earth and tested to see what different colors it makes over a Bunsen Burner. What I learned from this was, that Potassium (k) makes the color purple, Lithium Chloride (Li) turns the fire to the color red, Strontium Carbonate (Sr) makes the fire turn the color red/orange, Cupric Sulfate (Cv) makes the color green, Sodium Chloride (Na) turns the fire to the color orange, Boron (B) makes the fire turn the color green, Magnesium (Mo) makes the color white, Iron (Fe) turn the color orange. This all happens because the fire is reacting to all the chemicals and minerals that I had just previously mentioned. This information may be used to answer the question, "whats the effect of energy on elements?" What I believe went well was that we made the reactions happen, we took all the materials and had a pretty successful experiment. The problems were that first, we had to change Bunsen Burners because the original one sucked at it's job, second there was residue from previous flame lab experiments so that kinda interfered with our experiment, third we had the lights on so we couldn't really see the fire change color until we turned the lights off. For this experiment I think my group and I performed really well, we did have some setbacks but it didn't affect our group in total. If I had a second chance I would test the Bunsen burners before the day of the experiment to which is the best one. Then on the day of the experiment I would all the lights off so I could see the fire change colors better. I wouldn't change or trade my group in for anybody those group of people are the same people I would do the experiment 100 times over and over again. One way of continuing the experiment is making all the flames into a fire tornado and see how that works. And see how all the different color flames combined spinning and twirling, what type of color it would be i'm guessing black that would be awesome.








 RSS Feed
RSS Feed
