For this lab we created functioning batteries using pennies and cardboard soaked in vinegar.
Basic concepts
The first thing we did was take about six pennies and sanded off, one side of the pennies until it looked like the color silver. Now It make look like silver but it's not it's actually Zinc, so now one side of the penny should be copper and the other Zinc. Then after we sanded the pennies we then take a piece of cardboard and cut it into the shape of the penny. We then dropped our cardboard cut outs into a beaker of vinegar and let them soak while that was happening we took the pennies and put them on the small piece of aluminum foil. With the copper side of the penny facing down and the zinc side facing up, we then took a piece a soaked cardboard and dabbed it a little on a paper towel, after that we then applied on top of the penny (Zinc side facing up). After that we stacked our materials in a order that went copper, zinc, cardboard, you can repeat this pattern as many times as you would like but one end must have zinc facing out and the other should have copper. after that we tested the battery by connecting a L.E.D to it. At a molecular level electrons are jumping between the zinc, copper and vinegar in the cardboard. That flow of electrons is what creates the electric current that powers the L.E.D. Something I take away from this lab is to always persevere and never give up, my battery at first did not work but after a few trail and errors and with help from my partners I finally figured out how to do it.
Basic concepts
- Electricity is just electrons going through a wire
- All elements have electrons so as long as you can move the electrons in one atom to another you have electricity
- Some elements lose electrons more easily than others
- Pennies made past 1982
- Vinegar
- Cardboard
- L.E.D
The first thing we did was take about six pennies and sanded off, one side of the pennies until it looked like the color silver. Now It make look like silver but it's not it's actually Zinc, so now one side of the penny should be copper and the other Zinc. Then after we sanded the pennies we then take a piece of cardboard and cut it into the shape of the penny. We then dropped our cardboard cut outs into a beaker of vinegar and let them soak while that was happening we took the pennies and put them on the small piece of aluminum foil. With the copper side of the penny facing down and the zinc side facing up, we then took a piece a soaked cardboard and dabbed it a little on a paper towel, after that we then applied on top of the penny (Zinc side facing up). After that we stacked our materials in a order that went copper, zinc, cardboard, you can repeat this pattern as many times as you would like but one end must have zinc facing out and the other should have copper. after that we tested the battery by connecting a L.E.D to it. At a molecular level electrons are jumping between the zinc, copper and vinegar in the cardboard. That flow of electrons is what creates the electric current that powers the L.E.D. Something I take away from this lab is to always persevere and never give up, my battery at first did not work but after a few trail and errors and with help from my partners I finally figured out how to do it.
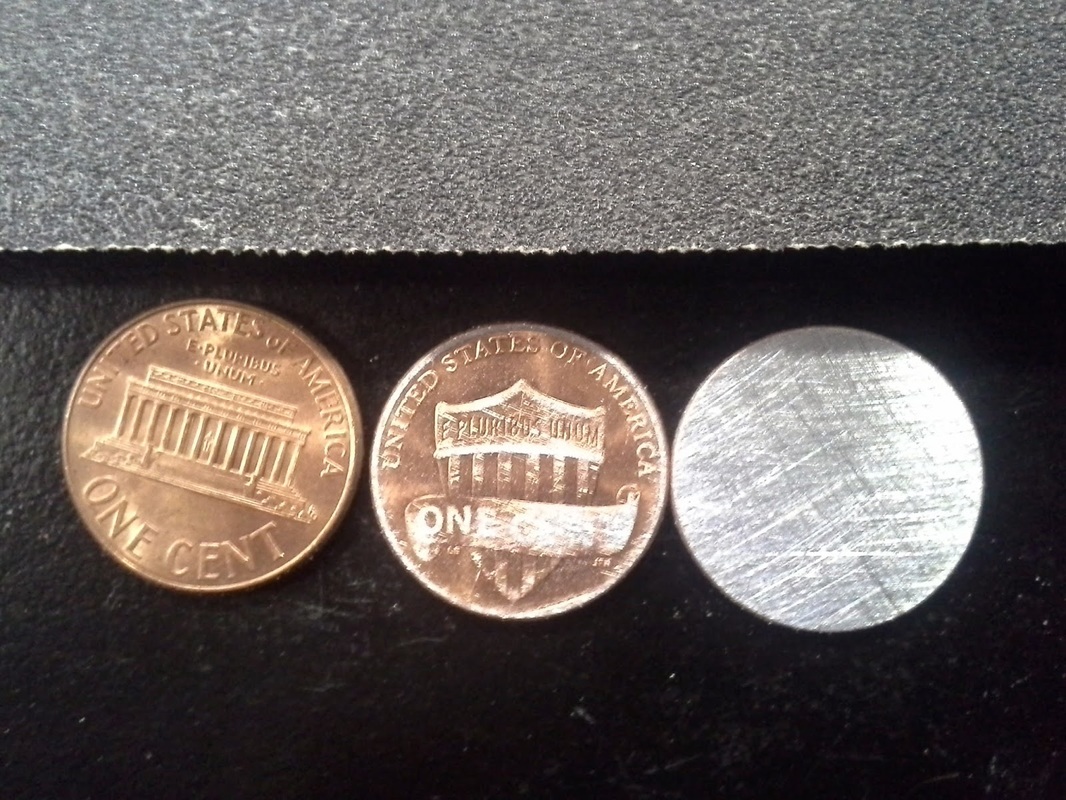


 RSS Feed
RSS Feed
